Without surgery, most patients die in childhood with a rate of survival of 66% at 1 year of age, 40% at 3 years, 11% at 20 years and 3% at 40 years1.
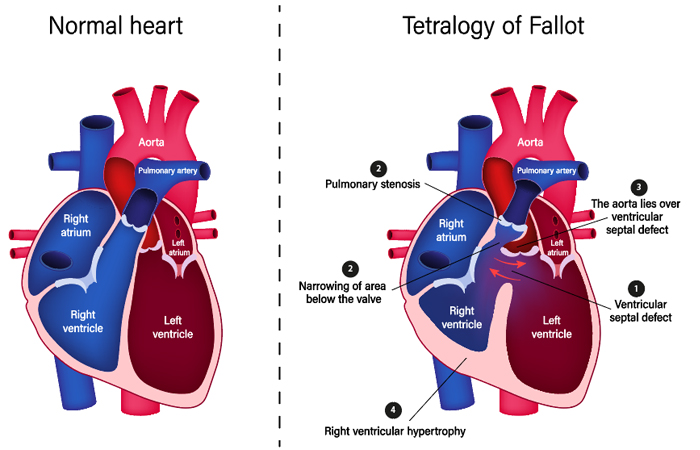
Less oxygenated “blue” blood returning to the right side of the heart is mixed with the oxygenated “red” blood from the left side of the heart through the hole and the overriding aorta. Babies with unrepaired TOF are often blue. Sometimes the pulmonary valve is also completely obstructed (pulmonary atresia). Other associated problems can include an atrial septal defect (hole between the two upper heart chambers) and abnormalities of coronary arteries (which supplies blood to the heart muscles).
Treating ‘Blue Baby'
In early infancy, babies with TOF may need a procedure to temporarily increase blood flow to the lungs (Blalock-Taussig shunt). Total surgical repair will be done later when the baby is older. With advances in surgery, survival into adulthood is expected, although repeat procedures are common.
Common problems which can remain in adults after repair include:
- Residual pulmonary valve narrowing or leaking. Also known as pulmonary valve stenosis or regurgitation
- Branch pulmonary artery narrowing
- Abnormal heart rhythms (arrhythmias)
Pulmonary valve regurgitation is very common in TOF patients with over 80% having at least moderate to severe pulmonary regurgitation2. A pulmonary valve replacement may be required depending on the patient’s symptoms, right ventricle enlargement, function and presence of arrhythmias. The options include surgical replacement or percutaneous pulmonary valve implantation (PPVI).
PPVI is a minimally invasive procedure to correct pulmonary valve stenosis or regurgitation. Only selected patients may be suitable for PPVI if they have a previous pulmonary valve replacement or conduit (tube replacement). It is performed in the cardiac catheterisation laboratory and the patient will be given general anaesthesia. The patient will lie on an X-ray table and an X-ray camera will move over the patient’s chest during the procedure. This allows the cardiologist to insert a thin tube (catheter) into a vein, typically in the leg and guide it to the heart. A deflated balloon is placed through this catheter and into the opening of the narrowed valve or conduit. The balloon is then inflated, which pushes the valve or conduit open so the blood will flow better.
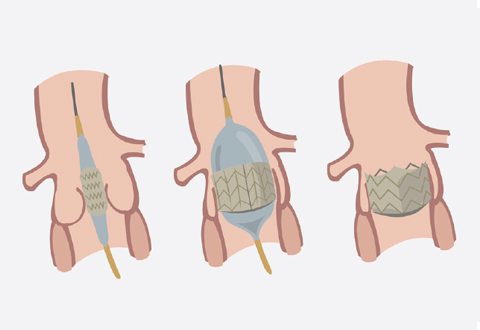
In PPVI, the deflated balloon is inflated to open up the artificial valve into position in the diseased valve.
Subsequently, an artificial heart valve attached to a wire, mesh-like tube (stent) is placed on the catheter and guided to the pulmonary valve. Once in place, the balloon is inflated to open up the valve into position. The catheter is removed from the body and the artificial heart valve becomes the new pulmonary valve.
The long term 10 year results3 of PPVI have been excellent and comparable to open-heart surgery, with 79% of patients not requiring reoperation. This is consistent with our experience at National Heart Centre Singapore, where the initial patients with TOF who had undergone PPVI have presented favourable five year results so far. The major advantage of this method over open-heart surgery is the shorter hospital stay and faster recovery time. Major guidelines4 have recommended PPVI over surgery for eligible patients.
1 Life expectancy without surgery in tetralogy of Fallot. E G Bertranou, E H Blackstone, J B Hazelrig, M E Turner, J W Kirklin. American Journal of Cardiology 1978;42:458-66
2 Late complications Following Tetralogy of Fallot Repair: The Need for Long-term Follow-Up. Ho KW, Tan RS, Wong KY, Tan TH, Shankar S, Tan JL. Annals of the Academy of Medicine, Singapore, 01 Nov 2007, 36(11):947-953
3 Jones TK, McElhinney DB, Vincent JA, Hellenbrand WE, Cheatham JP, Berman DP, Zahn EM, Khan DM, Rhodes JF Jr, Weng S, Bergersen LJ. Long-Term Outcomes After Melody Transcatheter Pulmonary Valve Replacement in the US Investigational Device Exemption Trial. Circ Cardiovasc Interv. 2022 Jan;15(1):e010852
4 2020 ESC Guidelines for the management of Adult Congenital Heart Disease
This
article is from Murmurs Issue 43 (May – August 2022). Click here to read other articles or issues.
