PARKINSON’S DISEASE (PD) is a chronic and degenerative brain disorder that results in motor impairment. After 4 to 6 years of treatment, medication-induced complications can develop. As the disease advances, symptoms become more severe and other problems related to the patient’s gait, cognition and musculoskeletal system develop.
In Singapore, PD is the second most common neurological disorder. Local studies have shown that the disease occurs in three out of every 1,000 people aged 50 and above. It is estimated that there are between 4,000 to 5,000 PD patients in Singapore [1].
Oral medication is the standard of care in early PD; however, as the disease progresses and medication-induced complications develop, patients may become refractory to this treatment option.
Deep Brain Stimulation (DBS) is the most common surgical treatment performed on people with PD, when medications alone are no longer able to adequately control their motor symptoms. It is also required when medication-related problems, particularly levodopa-induced dyskinesia (involuntary movements) and motor fluctuations, significantly affect the quality of life of the patient.
DBS therapy is an adjustable and reversible surgical treatment and more than 140,000 patients worldwide have been treated with DBS. In Singapore, more than 100 patients have benefited from DBS therapy. DBS has been approved for the following indications:
- Essential tremor
- Parkinson’s disease
- Primary dystonia
- Obsessive-compulsive disorder
- Epilepsy
SYMPTOMS OF PARKINSON’S DISEASE
PD is clinically defined by the presence of bradykinesia (slowness in movement), with at least one cardinal motor feature (rigidity or rest tremor).
In addition to the cardinal motor features, patients with PD also have non-motor symptoms involving a multitude of functions, such as disorders of the sleep–wake cycle regulation, cognitive impairment, disorders of mood and affect, autonomic dysfunction (mainly orthostatic hypotension, urogenital dysfunction, constipation and hyperhidrosis), as well as sensory symptoms (most prominently hyposmia) and pain
[2].

BENEFITS OF DBS THERAPY
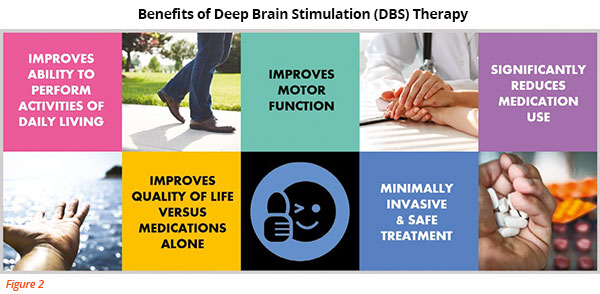
DBS therapy can give PD patients an additional 5 hours of good movement control per day, compared to the best medical therapy (using only oral medication). This can help the patient to regain the ability to perform normal daily activities and to improve their quality of life.
WHO SHOULD AND WHEN TO GO FOR DEEP BRAIN STIMULATION?
In February 2013, the EARLYSTIM study published in The New England Journal of Medicine provided Class I evidence for the use of DBS therapy in the earlier stages of Parkinson’s disease.
The trial recruited DBS patients in the early stages of PD (disease duration of ≥4 years and disease severity below 3 on the Hoehn and Yahr scale) and showed that DBS provided superior benefits for patients with early motor complications from PD, as compared to those who received only the best medical therapy
[3].
These findings led to an FDA approval in 2016, for DBS in patients with at least 4 years of disease duration and 4 months of motor complications, as an adjunct therapy for patients who are not adequately controlled with medications.
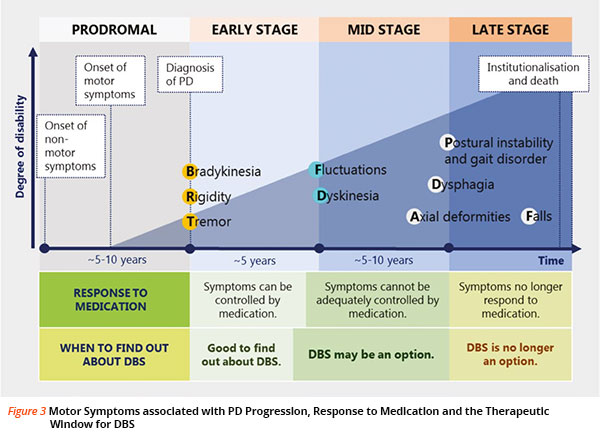
Deep brain stimulation for PD has a window of opportunity, where it may be most effective for PD patients. The window opens when a patient has the disease for at least 4 years’ duration that is not adequately controlled by medication, including motor complications for at least 4 months or of longer-standing duration.
Typically, the patient may present with one or more of the following symptoms observed:
- “On” time characterised by disabling dyskinesias (or other non-motor side effects)
- “Off” time characterised by disabling tremor, rigidity, or akinesia/bradykinesia
- Unpredictable motor fluctuations
- Medication-resistant tremor
The window of opportunity for DBS closes when:
- Symptoms no longer respond to dopaminergic medication
- The patient is severely disabled, even in the best “on” state
- Medical conditions prevent surgery
- The patient has dementia
(Refer to Figure 2)
DBS is not a cure for PD and it may not be suitable for everyone. Hence, people with PD will need to undergo a set of comprehensive tests and evaluations to assess their suitability before proceeding to a surgery.
Refer to Figure 3 for an overview of the disease progression and the therapeutic window.
Careful patient selection is performed by a multidisciplinary approach (comprising of neurologists, specialist nurses, neurosurgeons, anaesthetists, speech therapists, occupational therapists, physiotherapists, psychiatrists, psychologists and social workers). After surgery, once the device is turned on, our patients experience an immediate improvement in their movement. This leads to a significant benefit in their overall function and quality of life.
DBS patient support group sessions in the hospitals are held regularly to provide a platform for the PD community to share their experiences, and to provide insights and support.
HOW DOES DBS THERAPY WORK?
The exact mechanisms of how focal stimulation by DBS exerts local and systemic effects across the brain networks remain unclear. Initial views were based on the classic “rate model”, where the motor symptoms of PD were attributed to altered neuronal firing in the basal ganglia, wherein DBS creates a reversible “functional” lesion or a physiological block.
More recent models suggest that DBS acts through multifactorial mechanisms, including immediate neuromodulatory effects, synaptic plasticity and long-term neuronal reorganisation
[4,5].
WHAT IS IN A DBS THERAPY SYSTEM?
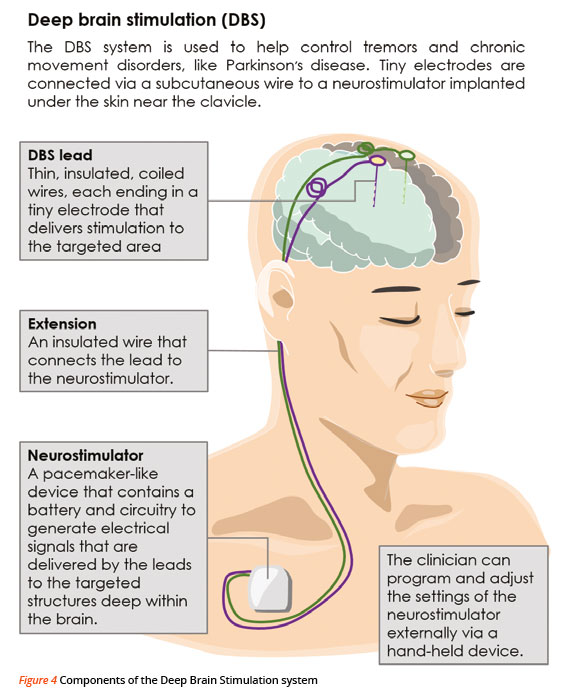
A small, pacemaker-like device is surgically placed under the skin, just beneath the collarbone. The device sends electrical signals through lead wires to a precise location in the brain, in either the subthalamic nucleus (STN) or globus pallidus interna (GPi) that controls movement. These signals block some of the brain’s messages that cause the annoying and disabling motor symptoms associated with PD. (Refer to Figure 4)
HOW IS DBS SURGERY PERFORMED?
DBS surgery is a minimally invasive procedure. Before the surgery, the patient is required to go for a brain MRI where the MRI images will be used on the day of the operation.
The surgery consists of 2 stages. In the first stage, a head frame is attached to the patient’s head and the purpose of the frame is to ensure that the head is immobile, so that the neurosurgeon can accurately and precisely place the DBS lead wires into the target location within the brain. The patient will go for a CT Scan with the head frame in place. The neurosurgeon then uses the pre-operative MRI and CT image to plan the best trajectory for the entry point of the lead electrodes.
Before the leads are placed permanently, several steps are taken to ensure that the permanent electrode is placed in the optimal position. A test electrode is used to locate the best spot in the brain. The location of the test electrode is checked by listening to the brain through microelectrode recording (MER; Refer to Figure 5). Besides MER, the patient under sedation will be woken up and asked to perform simple motor tests, such as moving their limbs or talking.
In the second stage, the patient will undergo the insertion of the pulse generator (or battery). Typically, the length of the hospital stay for a DBS surgery is 5 to 7 days.
The DBS battery will be turned on approximately 1 month after the surgery. Over the next 3 to 6 months, the patient will be followed up closely by the neurologist or APN for programming to find the best stimulation settings for the patient. In most cases, after programming, the patient’s PD medications will be reduced.

SUMMARY
As PD progresses at different rates for everyone, it is important to consider all the treatment options at the early stages of the disease.
By considering and starting DBS therapy early and at the appropriate time, patients with PD can experience greater therapeutic benefits and a better quality of life. Delaying DBS therapy might result in missing that window of opportunity altogether.
GPs can call for appointments through the GP Appointment Hotline at 6357 7095 or scan the QR code for more information.
By:
Assistant Professor Nicolas Kon Kam King, Consultant, Department of Neurosurgery,
National Neuroscience Institute (NNI)
Assistant Professor Nicolas Kon Kam King is a Consultant at the Department of Neurosurgery at the
National Neuroscience Institute (NNI), where he subspecialises in Functional Neurosurgery, Deep Brain Stimulation and Neuro-Oncology. He practises at both the Singapore General Hospital and the Tan Tock Seng Hospital campuses of the NNI. He is also the Co-Director of the NNI Deep Brain Stimulation programme.
REFERENCE
1. Tan, L.C.S., Venketasubramanian, N., Hong, C.Y., Sahadevan, S., Chin, J.J., Krishnamoorthy, E.S., Tan, A.K.Y. and Saw, S.M., 2004. Prevalence of Parkinson’s disease in Singapore Chinese vs Malays vs Indians. Neurology, 62(11), pp.1999-2004.
2. Poewe, W., Seppi, K., Tanner, C.M., Halliday, G.M., Brundin, P., Volkmann, J., Schrag, A.E. and Lang, A.E., 2017. Parkinson’s disease. Nature Reviews Disease Primers, 3, p.17013.
3. Schuepbach, W.M.M., Rau, J., Knudsen, K., Volkmann, J., Krack, P., Timmermann, L., Hälbig, T.D., Hesekamp, H., Navarro, S.M., Meier, N. and Falk, D., 2013. Neurostimulation for Parkinson’s disease with early motor complications. New England Journal of Medicine, 368(7), pp.610-622.
4. Lozano, A.M. and Lipsman, N., 2013. Probing and regulating dysfunctional circuits using deep brain stimulation. Neuron, 77(3), pp.406-424.
5. Ashkan, K., Rogers, P., Bergman, H. and Ughratdar, I., 2017. Insights into the mechanisms of deep brain stimulation. Nature Reviews Neurology.
