With ongoing advancements in cell-based therapies for cartilage repair, we can look forward to more accessible treatments and better patient outcomes for knee osteoarthritis and chondral defects of the knee. Find out more about the promising developments on the horizon and what they could mean for patients.
INTRODUCTION
The treatment of articular chondral defects of the knee has classically been challenging, due to the cartilage's aneural, avascular and alymphatic nature. This makes natural healing of chondral defects almost impossible.
CAUSES OF CHONDRAL DEFECTS OF THE KNEE
Patients can suffer from chondral defects from two main knee conditions: knee osteoarthritis and acute focal chondral injury.
1. Knee osteoarthritis
Age
This generally occurs in the older age group of patients 45 years and above.
Causes
Osteoarthritis is the gradual wear of the cartilage due to various factors such as:
- Biomechanical factors with malalignment being the most common
- Previous injury factors such as previous meniscus injuries which reduce shock absorption on the cartilage
- Inflammatory factors such as gout and rheumatoid arthritis
Presentation
The wear of the cartilage is generalised and chondral defects can occur from minor trauma, such as light jogging or twisting injuries, leading to a chondral flap tear and resultant defect when the tear detaches.
The defect side causes acute joint pain and swelling, and patients have pain on weightbearing and stair climbing.
2. Acute focal chondral injury
Age
This generally occurs in the younger age group of patients from paediatric to 45 years.
Causes
There is no generalised wear of the cartilage. Instead, a focal chondral defect is created due to an acute injury such as a sporting injury (e.g., anterior cruciate ligament tear via twisting injury, patella dislocation, landing from a jump of significant height).
Presentation
The chondral defect may be accompanied by bone loss, leading to an osteochondral defect. In addition to the symptoms of knee osteoarthritis, the detached fragment may be a loose body and might cause locking of the knee joint.
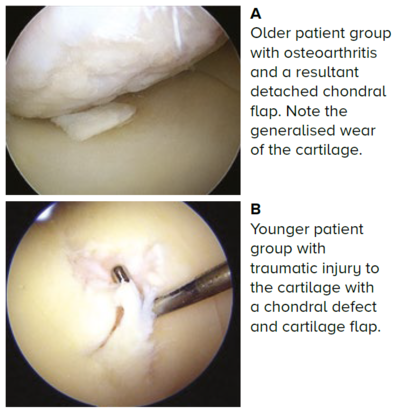
Figure 1 Arthroscopic pictures of chondral injuries in two different groups of patients
CURRENT ROUTINE CARTILAGE REPAIR STRATEGIES AND THEIR LIMITATIONS
Cartilage regeneration strategies can be broadly divided into non-cell-based therapy versus cell-based therapy.
1. Non-cell-based therapy
The gold standard for non-cell-based therapy is that of intrinsic repair enhancement.
What it is
With the use of microfracture, the surgeon creates holes through the tidemark of the subchondral bone after debriding the nonviable cartilage. This allows for marrow stimulation which will form blood clots, which in turn will mature into fibrocartilage to fill up the defect.
Limitations
This, of course, is inferior to that of native articular cartilage.
2. Cell-based therapy
Cell-based therapies include the gold-standard autologous chondrocyte implantation (ACI), a two-stage surgery.
What it is
In the first stage, the surgeon harvests healthy cartilage and sends it to the laboratory for culture. Thereafter, the cultured chondrocytes are embedded into a synthetic scaffold matrix. The cell-embedded matrix is then used for the second stage of surgery, where the chondral defect is debrided and the matrix is secured to the base of the debrided chondral defect via tissue glue.
Limitations
However, this method is costly, requires two surgeries, and is associated with donor site morbidity and has largely been avoided.1
A LATEST SURGICAL OPTION: AUTOLOGOUS MATRIX-INDUCED CHONDROGENESIS
Apart from ACI, which is available in Singapore, surgeons are advocating for a surgical procedure that requires only one surgery with minimal donor site morbidity.
Autologous matrix-induced chondrogenesis (AMIC) has thus been popularised amongst surgeons for exactly that. This surgery involves the use of a synthetically-created scaffold that aids in the regeneration of cartilage with the help of marrow stimulation techniques such as microfracture or chondroplasty.
To improve the biological aspects of repair, many surgeons advocate for the use of bone marrow aspirate concentrate (BMAC) which contains bone marrow mesenchymal stem cells (BM-MSC) in small concentrations along with growth factors and cytokines.2
A single-step procedure
The surgeon would harvest BMAC from the iliac crest and spin it down to a concentrate. Thereafter, the BMAC will be embedded into the scaffold and then grafted to the debrided chondral defect as a one-stage surgery, avoiding the complications of ACI.
Results from randomised controlled trials have shown comparable results at two years between ACI and AMIC.3
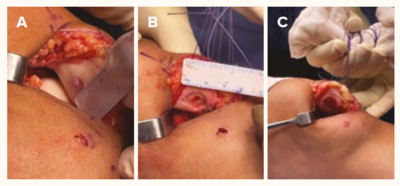
Figure 2 The surgical procedure of AMIC
A. Chondral defect of the patella
B. Debridement of the unhealthy cartilage to stable edges
C. AMIC and BMAC scaffold application
FUTURE POTENTIAL CELL-BASED THERAPIES FOR CARTILAGE REGENERATION
1. Autologous bone marrow mesenchymal stem cells (BM-MSC)
To circumvent the need for two surgeries and to improve the clinical outcomes of cartilage regeneration, the search for an alternative cell source has been ongoing for the last 10 years.
A suitable candidate to substitute ACI chondrocytes was autologous BM-MSC, which are bone marrow stem cells harvested from the patient's own bone marrow.
Benefits
Researchers in Singapore have shown that concentrated autologous BM-MSC in high dosages (without scaffold), at 10 years follow-up in clinical trials:4
- Was as effective
- Required one less surgery
- Was cheaper
- Has much lesser donor site morbidity
For AMIC with BMAC, the results were more successful for patients under 45 years old.
However, with concentrated BM-MSC alone, there were no significant differences seen for patients at any age, indicating superiority in the use of concentrated BM-MSC.4 We hope to see this option available in routine clinical practice soon.
BM-MSC has also been investigated as an intra-articular injection for chondral defects with good results, and this is a potentially attractive option as it can be administered in the clinics.5
2. Other sources of stem cells
Other sources of stem cells being investigated for cartilage regeneration include adipose-derived stem cells and umbilical cord blood-derived stem cells, and there are clinical trials using these stem cells for intra-articular injections for osteoarthritis of the knee.
There has been evidence of improving pain and function of the knee in the short term, but the evidence is still limited in terms of actual cartilage regeneration and long-term results.6
3. Further in the future: 'Cell-free' cartilage regenerative therapy – stem cell exosomes
Limitations of autologous stem cells
Cell-based therapies such as autologous BM-MSC may have promising results, but autologous stem cells have limited cellular capacity for self-renewal, proliferation and differentiation with increasing donor age.
Cell-based therapies thus pose significant logistic and operational challenges associated with proper handling and cell storage to maintain cell viability and vitality.
Paradigm shift in understanding MSC therapy
There is increasing evidence that MSC therapy is not dependent on the engraftment of cells at the site of injury, followed by differentiation of the MSC to the target cell type.7
We used to believe that the stem cells, when injected to the injury site, will attach to the injury site and then differentiate into the target cell type to regenerate new tissue. However, this is not the case.
There has been a paradigm shift with evidence that MSC are signaling cells, or 'messenger signaling cells', that mediate the effect by secretion of trophic factors. These trophic factors then stimulate the intrinsic repair of the damaged tissues.
MSC exomes as 'cell-free' therapy
One of these important factors is the MSC exosome. MSC exosomes are nano-sized, cell-secreted bi-lipid membrane vesicles of about 40-100 nm present in the MSC secretome that have been found to possess potent immunomodulatory and regenerative properties.7
Animal studies have been performed and MSC exosomes alone without scaffold have demonstrated significant cartilage regeneration of articular hyaline cartilage, similar to native cartilage, with comparable histological and biomechanical properties.8
This 'cell-free' regenerative therapy, which is a clear liquid that can be commercialised and injected in clinics, is an exciting field to watch for the future of bringing cartilage regeneration into clinics.8
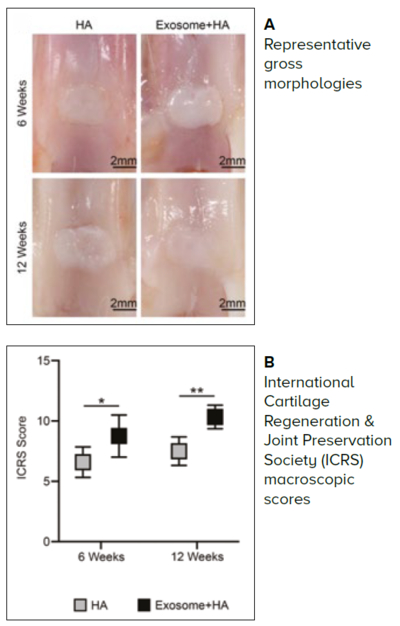
Figure 3 Animal studies showing superior cartilage regeneration with MSC exosomes and hyaluronic acid (HA), as compared to HA alone.8
Values represent the means ± 95% CI. *P < 0.05, **P < 0.01 compared to HA group. Scale bar: 2 mm.
CONCLUSION
Current cell-based therapies for cartilage regeneration involve the usage of AMIC and BMAC, but have less successful outcomes in patients above 45 years old. More advanced cellular therapy options for cartilage regeneration, such as concentrated forms of BM-MSC, have shown sufficient clinical evidence for future routine clinical practice and may be suitable for older patients above 45 years old.
The future of cartilage regeneration is exciting and may venture into cell-free therapy that can be delivered to the clinics, such as that of MSC exosomes.
REFERENCES
- Mistry H, Connock M, Pink J, Shyangdan D, Clar C, Royle P, Court R, Biant LC, Metcalfe A, Waugh N. Autologous chondrocyte implantation in the knee: systematic review and economic evaluation. Health Technol Assess. 2017 Feb;21(6):1-294. doi: 10.3310/hta21060. PMID: 28244303; PMCID: PMC5346885.
- Gobbi A, Whyte GP. Long-term Clinical Outcomes of One-Stage Cartilage Repair in the Knee With Hyaluronic Acid-Based Scaffold Embedded With Mesenchymal Stem Cells Sourced From Bone Marrow Aspirate Concentrate. Am J Sports Med. 2019 Jun;47(7):1621-1628. doi: 10.1177/0363546519845362. Epub 2019 May 16. PMID: 31095402.
- Fossum V, Hansen AK, Wilsgaard T, Knutsen G. Collagen-Covered Autologous Chondrocyte Implantation Versus Autologous Matrix-Induced Chondrogenesis: A Randomized Trial Comparing 2 Methods for Repair of Cartilage Defects of the Knee. Orthop J Sports Med. 2019 Sep 17;7(9):2325967119868212. doi: 10.1177/2325967119868212. PMID: 31555714; PMCID: PMC6749791.
- Teo AQA, Wong KL, Shen L, Lim JY, Toh WS, Lee EH, Hui JHP. Equivalent 10-Year Outcomes After Implantation of Autologous Bone Marrow-Derived Mesenchymal Stem Cells Versus Autologous Chondrocyte Implantation for Chondral Defects of the Knee. Am J Sports Med. 2019 Oct;47(12):2881-2887. doi: 10.1177/0363546519867933. Epub 2019 Aug 21. PMID: 31433674.
- Wong KL, Lee KB, Tai BC, Law P, Lee EH, Hui JH. Injectable cultured bone marrow-derived mesenchymal stem cells in varus knees with cartilage defects undergoing high tibial osteotomy: a prospective, randomized controlled clinical trial with 2 years' follow-up. Arthroscopy. 2013 Dec;29(12):2020-8. doi: 10.1016/j.arthro.2013.09.074. PMID: 24286801.
- Ha CW, Park YB, Kim SH, Lee HJ. Intra-articular Mesenchymal Stem Cells in Osteoarthritis of the Knee: A Systematic Review of Clinical Outcomes and Evidence of Cartilage Repair. Arthroscopy. 2019 Jan;35(1):277-288.e2. doi: 10.1016/j.arthro.2018.07.028. Epub 2018 Nov 16. PMID: 30455086.
- Lai RC, Tan SS, Teh BJ, Sze SK, Arslan F, de Kleijn DP, Choo A, Lim SK. Proteolytic Potential of the MSC Exosome Proteome: Implications for an Exosome-Mediated Delivery of Therapeutic Proteasome. Int J Proteomics. 2012;2012:971907. doi: 10.1155/2012/971907. Epub 2012 Jul 18. PMID: 22852084; PMCID: PMC3407643.
- Wong KL, Zhang S, Wang M, Ren X, Afizah H, Lai RC, Lim SK, Lee EH, Hui JHP, Toh WS. Intra-Articular Injections of Mesenchymal Stem Cell Exosomes and Hyaluronic Acid Improve Structural and Mechanical Properties of Repaired Cartilage in a Rabbit Model. Arthroscopy. 2020 Aug;36(8):2215-2228.e2. doi: 10.1016/j.arthro.2020.03.031. Epub 2020 Apr 14. PMID: 32302651.
Assistant Professor Francis Wong is a Consultant with the Department of Orthopaedic Surgery, Sengkang General Hospital, and Assistant Professor at Duke-NUS Medical School. He has completed a Masters of Clinical Investigation, and is pursuing a part-time PhD at the National University of Singapore via the National Medical Research Council Research Training Fellowship.
His clinical interests are in sports medicine and sports surgery, in particular cartilage regeneration and restoration, where he became the first Fellow of the International Cartilage Regeneration & Joint Preservation Society from South-East Asia.
GPs who would like more information on this topic, please contact Prof Wong at francis.wong.k.l@singhealth.com.sg.
GPs can contact the SingHealth Duke-NUS Cell Therapy Centre at sd.cell.therapy@singhealth.com.sg to know more about the available cell therapies and clinical trials on the SingHealth campus.
